Two days before Christmas I took my Dupixent (dupilumab) loading dose in an empty dermatology clinic room. I’ve resisted started a second biologic (I’m still taking Skryizi for psoriasis) for almost five years. The unknown of what two biologics would do to my body kept me from taking this plunge–until now.
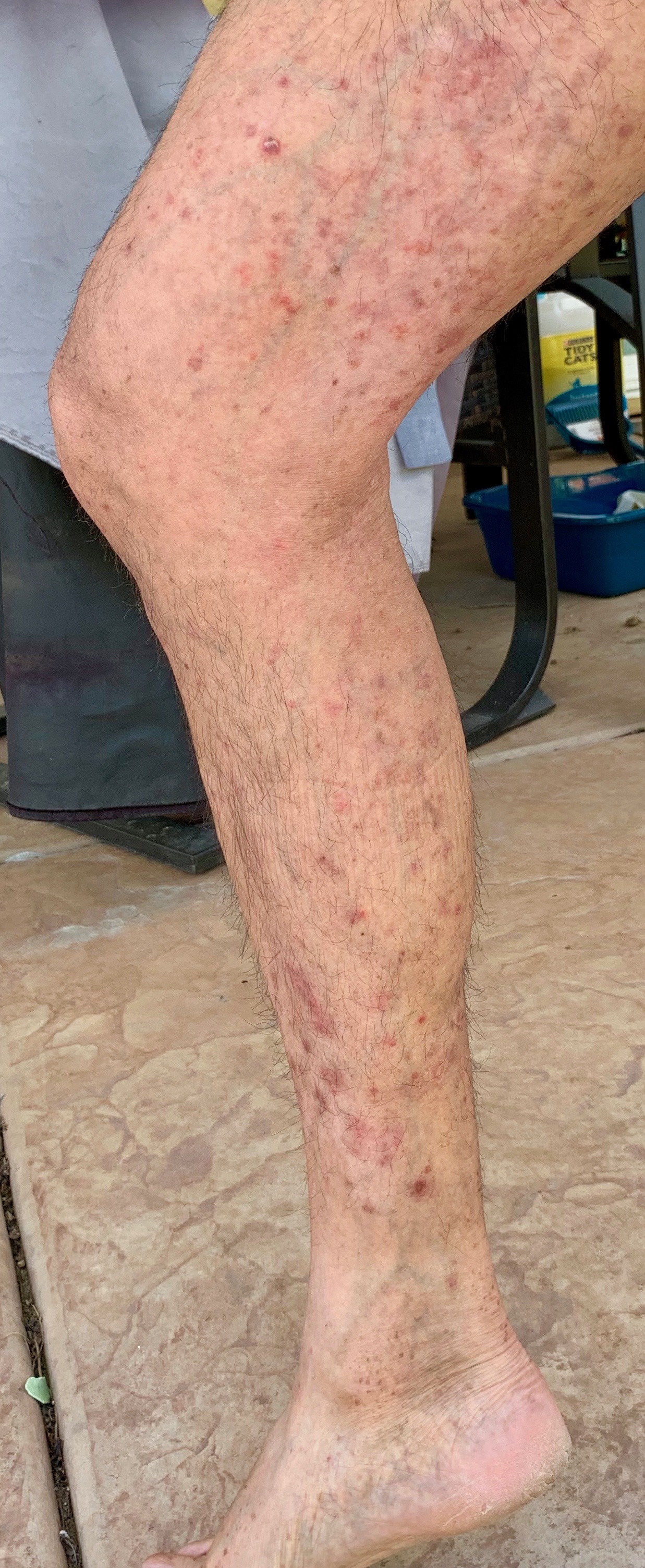
The last month I’ve had a miserable eczema flare. While I wanted to enjoy the holidays, I struggled mightily with rashes all over my body. The eczema on my back is the worst it’s been in a long long time. I’ve tried every treatment from a variety of topicals, moisturizers, wet wraps, antihistamines. I’ve modified my diet, increased exercise, and taken it easy at work. Not much has helped.
Hitting a Wall of Rashes
Dr. C believes that this eczema flare built up throughout the fall season. I traveled to the Bay Area a couple of times for meetings. Trips to Oregon and Dallas for conferences increased my exposure to allergens. The trip to Oregon, in particular felt triggering for my eczema and asthma.
The trip to Santa Cruz, about a 3 hour drive from my home, turned out great for work, but poor for health. I reacted (as usual) to the old hotel room that carried the smells of perfume drenched toiletries provided in the bathroom. Oregon is a beautiful place to visit if you love the outdoors and mountain views. What I didn’t enjoy on my trip to central Oregon was the dust, smoke, dry air (low humidity, and slightly scented sheets in my AirBnB bedroom. Here is a photo from downtown Bend, OR.

My skin also didn’t take well to my trip to Dallas where the hotel room had a musty smell to it and the Texas BBQ brought on a night of restlessness. Busy conference schedules, especially those early morning meetings after only sleeping for a few hours due to itchy wakefulness, stress my body and my skin.
By Thanksgiving I felt grateful for a busy travel season to end, but my immune system, decided to push back hard. The bumpy, itchy rash on my forearm, chest, and upper back serve as the proverbial canary in the coal mine warning me of much worse to come. Soon my forehead, scalp, trunk, and later, legs, joined the inflammation party.
Decided How to Treat the Flare
My standard go to treatment when eczema flares is wet wrap therapy. Here is how I describe it in my linked blog post:
The treatment is primarily for eczema, but it helps my psoriasis too. WWT involves bathing, slathering (my doctor’s word not mine) a medium strength topical corticosteroid ointment on my skin, then covering it with wet clothes. Over the wet clothes I put a layer of dry clothes which helps keep the wet clothes damp. After about an hour I re-wet the wet layer of clothes. Some leave the wet clothes on overnight, or for some hours. I usually leave it for about two to three hours in the evening. The treatment moisturizes the skin, allows the medicine to penetrate the skin, and cools inflammation.
I tried wet wrapping a number of times, but the eczema only became more inflamed and angry. I felt at a loss what to do next. Waiting the flare out sometimes works, but can take weeks. So, I contacted Dr. C for a consult. Thankfully, she had an appointment for me in a couple of days.
Dr. C confirmed the flare is primarily eczema. Skyrizi is managing psoriasis quite well after five years amazingly. She offered systemic steroids pills as a way to suppress the inflammation, but I balked at the idea. For decades doctors told me that systemic steroids could severely flare psoriasis.
She then thought we should put Dupixent back on the table as an option. I’ve felt concerned about taking two biologics concurrently. Would insurance over the cost? Would it flare psoriasis? My allergist pointed me to studies that showed Dupixent triggering new cases of psoriasis or exacerbating existing ones. That didn’t sound great.
I left that urgent appointment with just topical medications tacrolimus and Opzelura (ruxolotinib) to try instead of the medium strength topical steroid (TCS) treatment.
Will Dupixent Work?
With just over a week since the Dupixent loading dose I’m hoping it will kick in soon. The itchiness at night is still really intense as I’ve only slept through the night a handful of times in the past month, and that’s with antihistamines.
I’m wary of whether or not it will trigger my psoriasis or existing eye issues (more on these in a coming post). Other potential side effects aren’t too far behind in my worries. For now we’ll wait and see.