Beautiful flowers in Sitka, Alaska, taken during a stop on our summer Alaska cruise vacation with family.
A couple weeks ago marked the 42nd week on Tremfya (guselkumab) and the 7th injection. I started in mid-August last year with about ten months elapsed since that first injection on the day of solar eclipse in August.
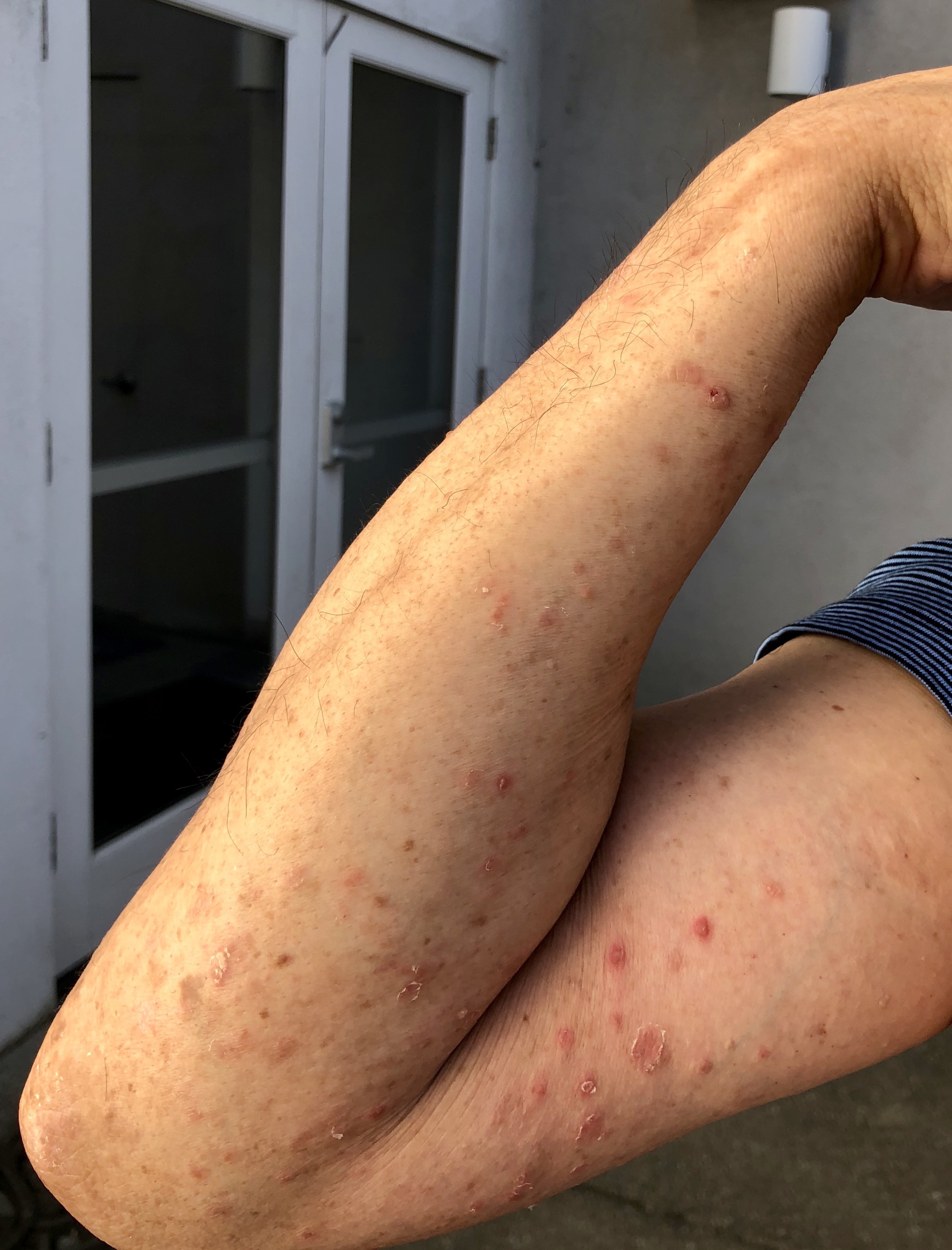
At this point in my treatment cycle I hoped to have more stability with my skin. But skin flares do happen even while taking the best medications.
What a Bad Skin Flare
Somehow I think that if I’m taking a super expensive biologic for psoriasis that I’m entitled to clear skin. Expectations often wreak havoc on my mood when I think this way. As an optimist and idealist it’s quite easy to bring those unrealistic expectations to the new medications I take.
When my skin flared like a wind swept wildfire a month ago I felt overwhelmed. Between the darker red spreading psoriasis spots and the generalized bumpy rash all over my body I could only feel normal skin in a few places. My mood darkened as I went into coping mode.
When my skin flared like a wind swept wildfire a month ago I felt overwhelmed.
A few factors appeared to contribute to the flare: increasing phototherapy dosage too quickly; Tremfya losing effectiveness at the end of the 8 week cycle; completely stopping cyclosporine; stress at work with a demanding spring; and a busy schedule with end of school year activities for the kids.
Tremfya (Guselkumab) Week One
3 Weeks with Tremfya: The Waiting Game
8 Weeks with Tremfya: It’s Working!
12 Weeks with Tremfya: The Third Injection
16 Weeks with Tremfya: The Verdict?
20 Weeks with Tremfya: Read the Instructions! (4th Injection)
24 Weeks with Tremfya: A Pattern Emerges
28 Weeks with Tremfya: Still Working? (Injection #5)
38 Weeks with Tremfya: The Q&A Edition
Employing Poor Coping Strategies
I’m not proud to report that during the flare I didn’t cope well at all. I did continue my skin care routines including taking a break from phototherapy and using steroids with wet wraps a couple times. But my expanding gut tells me that I ate way too much junk food. All those pastries, ice cream, donuts, extra shots of espresso, greasy burgers, and chips of all sorts no doubt made my skin even worse.
I also tend to stay up later when my skin flares. In an ineffective attempt at avoiding caring for my skin I delay my shower and night time skin routines. Instead I play games on my cell phone, (binge) watch Netflix shows, work on emails, or read online sports news. Eventually I get going on what I need to do, but much later than I’d like. The next day I’m naturally tired and wanting naps in the afternoon.
I really could use more margin in my life for when difficulties like skin flares inevitably come. It’s not that I don’t know better ways to cope when my health tanks. But if I’m already tired and stressed I don’t have the will nor the energy to counter those cravings to eat junk food and stay up late.
I really could use more margin in my life for when difficulties like skin flares inevitably come.
Thankfully, vacation came at a great time. A week on an Alaska cruise settled my mind, gave me much needed time away from work, and allowed the new Tremfya injection to start working.

Waterfall next to Mendenhall Glacier in Juneau, Alaska
Treatment: A Work in Progress
I hear stories of how long it takes to finally find a treatment plan that effectively manages psoriasis while minimizes risk. Some people go through multiple treatments, including combinations of various medications such as topical, phototherapy, systemic pills, and injectable biologics.
I feel like I’ve been trying to find that elusive treatment plan for the past twelve years. When the systemic Soriatane with phototherapy stopped working for me in the mid-2000s my skin would not stop peeling and scaling. Pure misery. I tried new biologics, went back to methotrexate, and even tried coal tar. Nothing worked.
Cyclosporine turned out to be the most effective medication for my psoriasis and eczema. However, it is only FDA approved for psoriasis for one year because of it’s toxicity to the kidney and other side effects. I used cyclosporine off and on for the next decade while trying each new biologic, although the breaks would only last a couple months before I needed to start up again.
Psoriasis treatment just feels like a work in progress, especially when that bad flare comes along unexpectedly.
Now I’ve finally stopped cyclosporine while taking Tremfya and experimenting with phototherapy. Psoriasis treatment just feels like a work in progress, especially when that bad flare comes along unexpectedly.
Time to end this entry with a photo of a cool glacier in Alaska to remember those calmer days. Too bad I can’t just go back here whenever my skin flares!

Glacier Bay National Park, Alaska